In the context of integrated pest and disease management for fruit trees, and cherries in particular, the most relevant tool is monitoring. Its objectives are: to detect the presence and abundance of pests and natural enemies early, to determine the life stages present (eggs, larvae, nymphs, pupae, and/or adults), the location of insects and mites on the plant, and their distribution within the orchard.
All information obtained from monitoring is vital for making informed pest management decisions.
Managing information about the life stage of pests allows us to determine when they are most susceptible, leading to more effective control measures. Furthermore, pest monitoring should be complemented with orchard information, such as the crop’s phenological stage, the presence of weeds and other surrounding flora, and the arthropods that may be associated with these plants.
Another important aspect is that monitoring insects or other types of pests not only helps prevent these problems. In certain cases, it also complements disease management. For example, the presence of certain insects like scale insects or bark beetles facilitates the entry of wood-decay fungi.
Others, such as Drosophila suzukii, carry yeasts and bacteria that initiate sour rot in fruits, especially cherries. Therefore, monitoring as an integrated management tool based on control is absolutely necessary for the development of sustainable orchards.
Among the important aspects to consider for effective monitoring are:
Having trained personnel with the necessary equipment (at least a 10x magnifying glass and data collection forms). Perform this monitoring periodically, following a protocol tailored to the orchard.
It is important to keep records of field data and analyze them to make timely and appropriate management decisions.
The following are the economically important pests associated with cherry trees, along with some aspects to consider for effective monitoring.
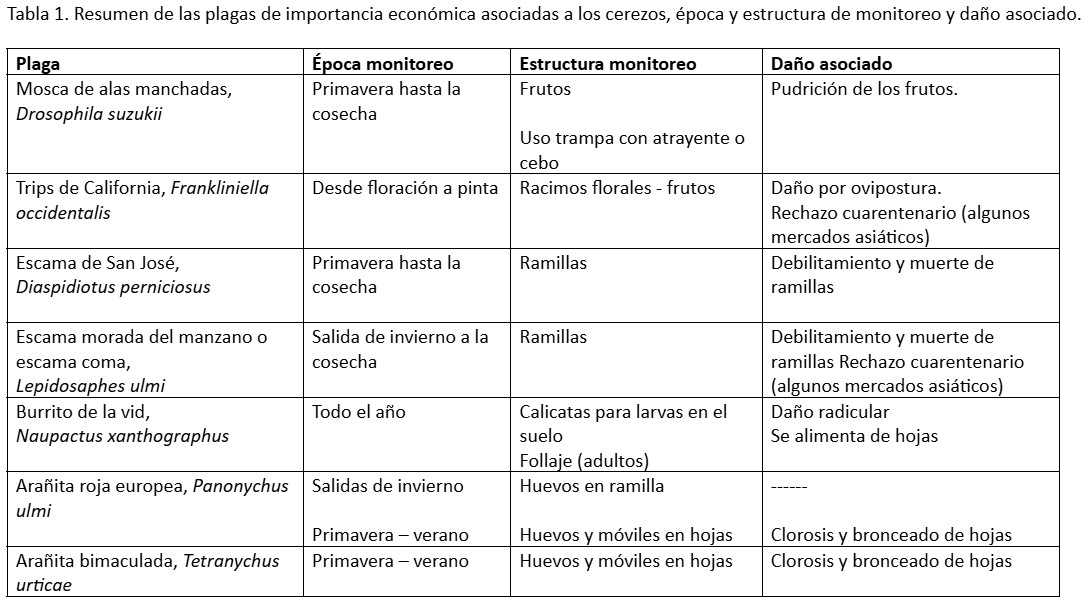
Spotted wing drosophila: Drosophila suzukii
The critical period for Drosophila suzukii infestation (Photo 1) is when it lays its eggs on the fruit (Photo 2), from the straw-colored stage onward, for 2 to 3 weeks.
The most efficient monitoring method is fruit inspection. It is suggested to randomly inspect approximately 100 fruits per hectare, a relatively quick process that provides a very good approximation of the pest’s status.
In addition, traps with fermentation attractants or synthetic baits can be used from the beginning of the season (September) for early detection in the area.


Western fruit thrips: Frankliniella occidentalis
The most important monitoring period for Western Fruit Thrips (Photo 3) is between flowering and fruit set. It is important to determine thrips densities in the flower clusters from the beginning of flowering in order to make timely management decisions.
These thrips densities are monitored by shaking the flower clusters onto a sheet of paper, counting the insects that fall, and observing the fruit from fruit set onward. In each orchard, it is suggested to sample 20 to 25 trees per orchard (4 to 5 flower clusters per tree), randomly distributed.


San Jose scale: Diaspidiotus perniciosus
Monitoring is carried out by inspecting twigs for migratory nymphs, adults, and live nymphs (Photograph 5). This scale has two to three generations of migratory nymphs, between October and November and April (depending on the production area). In each plot, it is suggested to sample 20 trees per plot, randomly distributed, inspecting 10 twigs per tree.
Additionally, pheromone traps (commercially available) can be used, which allow for the detection of male flights associated with nymph migration.

Purple scale or comma scale, Lepidosaphes beckii
This scale insect has at least two overlapping generations per season, meaning that all life stages can be found during the season (Photo 6). Monitoring for this scale is carried out on 1- to 3-year-old twigs, where the presence of live individuals of the different life stages should be determined. In each plot, it is suggested to sample 20 trees, examining 10 twigs on each tree.

Grapevine weevil, Naupactus xanthographus
Given the weevil’s behavior, where most of its life cycle is spent in the soil (from larva to adult), monitoring is carried out differently depending on the stage being monitored.
Larval monitoring (photo 7): Dig pits (10 pits per plot), each 35x35x45 cm deep, and record the number of larvae, pupae, and pre-emergent adults in each pit.
Adult monitoring (photo 8): This is done on the foliage, monitoring 15 to 20 plants per plot, randomly distributed. Traditionally, a 3 x 1.5 meter polyethylene sheet is placed on the ground on both sides of the plant to collect and count the adults that fall when the foliage is struck with a rubber mallet.


European red mite, Panonychus ulmi, and two-spotted spider mite, Tetranychus urticae
Monitoring for the European red mite should be carried out on twigs during winter outings to observe eggs and mobile stages (nymphs and adults) on the upper surface of the leaves, from spring onwards. During this same period (spring-summer), monitoring for the two-spotted spider mite should be performed by observing eggs and mobile stages on the leaves. For monitoring, at least 20 trees per plot should be inspected, observing 10 leaves and/or twigs (depending on the season and pest).
The following table indicates the plant structures, associated damage, and the most relevant monitoring periods for economically important pests associated with cherry trees.
Just like arthropod pests, pathogens must also be monitored to improve and optimize control strategies. Today, thanks to techniques like qPCR, we can determine the amount of inoculum present in critical tissues, such as flowers or fruit, almost in real time. This helps us understand if there is a risk of infection, where the probability of that inoculum developing into rot is high due to its abundance. This type of monitoring is carried out both pre- and post-harvest.
Currently, we are sampling fruit, starting at the straw-colored stage, to identify populations of Botrytis, Geotrichum, and Alternaria—all fungi capable of causing rot that leads to fruit rejection by our clients. In post-harvest, packing line surfaces, cold storage facilities, and, most importantly, the water used for cherry sorting must be frequently checked for the same fungi found in the field. In addition, we are also monitoring for Penicillium and yeasts as indicators of contamination.
The current season, due to an erratic spring, is particularly complex in phytopathological terms, so monitoring must be a mandatory part to establish a sustainable and high quality/condition production of our fruit.
Literature consulted:
- Devotto L. (2025). Drosophila suzukii: How to implement strategic management of its silent threat to cherry harvesting (interview). Available at:
https://smartcherry.cl/planificacion-tareas/control-de-lepidopteros-y-drosophila/drosophila-suzukii-como-realizar-un-manejo-estrategico-ante-su-amenaza-silenciosa-en-la-cosecha-de-cerezas/ - Fuentes E. (2021) Quarantine pests of cherry trees. Available at:
https://comitedecerezas.cl/wp-content/uploads/2021/09/8-Plagas-cuarentenarias-cerezo-Drosophila-suzukii-1.pdf - Fuentes E. (2024). Eduardo Fuentes: A look at the main pests of cherry trees. Available at:
- PDT Cerezos. (2010). Cherry Tree Pests and Diseases Guide. Available at: http://www.fdf.cl/biblioteca/publicaciones/2010/archivos/Guia_Plagas_Enf_Cerezos.pdf
- Prado E. (1990) Cherry Tree Pests. https://biblioteca.inia.cl/server/api/core/bitstreams/1dd20970-a335-4645-8ffb-234a5f11bad9/content
- Ripa, R., Luppichini, P. (2010). Walnut Pest Management. INIA Book Collection No. 25. Ministry of Agriculture. 116 p.
- Ripa, R., Luppichini, P. (2010). Vine Pest Management. INIA Book Collection No. 25. Ministry of Agriculture. 145 p.







