The best time to apply gibberellic acid to cherry trees is at the beginning of stage 3 of cell elongation. The beginning of stage 3 can be recognized by the color change from green to yellow, commonly known as straw color, which in practice coincides with the beginning of veraison.
Fruit size is undoubtedly one of the most important attributes in cherry production; quality and size are fundamental when exporting, as the financial returns received by producers and exporters depend on these and other factors combined.
Each season presents a constant challenge: producing high-quality, well-conditioned fruit. This means attractive fruit with good firmness, hydrated pedicels, no damage, and of a good size.
The sustained increase in cherry production, which reached over 125 million 5 kg boxes (equivalent to approximately 626,000 tons) during the 2024-2025 season, further underscores the importance of quality and maintaining this attribute throughout the supply chain. From a commercial perspective, despite the complexity of the 2024-2025 season, sizes ≥2J make all the difference.
A series of agronomic management practices can be implemented holistically, including proper adherence to nutritional programs, appropriate irrigation management, and thinning, among others, to improve the source-sink ratio and, consequently, fruit quality. This relationship can also be enhanced exogenously through the use of growth regulators, compounds that act as modulators of phytohormones and, when applied at key moments in fruit development, can influence processes directly related to fruit size, such as cell division and elongation.
Gibberellins
Phytohormones are grouped into nine main classes: auxins, gibberellins, cytokinins, ethylene, abscisic acid, brassinosteroids, jasmonates, and strigolactones, the latter recognized since 2012.
In this article, we will discuss the use of gibberellic acid or gibberellins (GA3) in cherry production, focusing on its application timing, dosage, and main physiological effects on fruit development.
Gibberellins were discovered through studies on the ascomycete fungus Gibberella fujikuroi, the causal agent of “bakanae” or abnormal elongation in rice. These substances are ditepenoid acids and are identified by the abbreviation GA (gibberellic acid), followed by a number indicating the order in which they were discovered and characterized. Among those with the greatest biological activity are GA1, GA3, GA4, GA5, GA6, and GA7, while the remaining ones act as precursors.
Gibberellic acid is one of the most studied phytohormones in plant physiology because it participates in processes such as cell division and elongation, shoot and fruit growth, internode elongation, promotion of flowering in certain annual species, inhibition of floral induction in many perennial species, and it promotes germination.
Currently, there are countless GAs (more than 100). However, only a few have significant biological activity. In cherry production, GA3 is the most widely used, with various objectives related to development, including shoot growth and enhancing fruit elongation in the final stage.
How does gibberellic acid increase fruit size?
Gibberellic acid acts at the cellular level by stimulating proton pumps (H+-ATPase) in the plasma membrane, causing protons (H+) to move into the cell wall, lowering its pH and creating an acidic environment. This activates proteins called “expansins,” which break the bonds between cellulose molecules, making the cell walls more elastic.
As a result, the influx of water then allows for cell elongation, leading to an increase in fruit size.
Fruit growth depends on the accumulation of dry matter and water, and is determined and eventually limited by the source-sink ratio and the availability of metabolites within the plant. The fruit’s sink capacity is a dominant factor in the competition for photoassimilates and is primarily defined by flower quality and the number of flowers produced on the tree (Guardiola, 1997).
Research and practical experience
Whiting & Lang (2021) demonstrated that in the Sweetheart and Lapins cultivars, applications of GA3 at concentrations between 20 and 40 ppm significantly increased fruit diameter and weight, shifting the size distribution toward larger sizes.
In Chile, various studies have described the effects of gibberellic acid on the quality and condition of cherry fruit. Carrión-Antolí and Zoffoli (2024) showed that this application affects the modulus of elasticity and resistance to mechanical damage postharvest. They also graphically demonstrated that GA3 applications significantly increase the fruit growth rate from stage 3 onward compared to a control treatment, resulting in a larger final fruit size at harvest.
Avium’s research and development team, in conjunction with cherry growers in Chile, has conducted a series of trials demonstrating the significant effect of using GA3 as a supplement to the nutritional program for cherry trees. These trials have shown improvements in soluble solids, dry matter, and firmness (Durofel), and especially in fruit size, with each of these parameters being directly and positively influenced by exogenous applications of GA3 (Tapia, C. 2015).
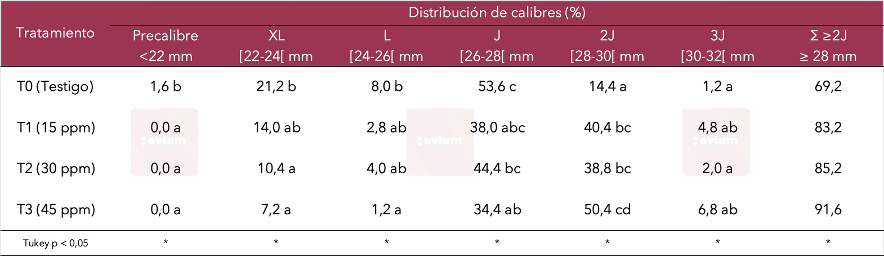
Furthermore, the effect of complementary GA3 applications has demonstrated a clear positive impact on average fruit size and weight, as well as a clear shift in the size distribution towards those with greater commercial appeal (Tables 1 and 2).
Table 1. Effects of GA3 application on the diameter and weight of Lapins fruit (Avium, 2015).

Table 2. Effects of GA3 application on the distribution of calibers cv. Lapins (Avium, 2015).
When and how much should GA3 be applied?
Over time, it has been established that the optimal time to apply gibberellic acid is precisely at the beginning of stage 3 of cell elongation. The start of this stage can be recognized by the fruit’s color change from green to yellow, commonly known as straw color. However, in practice, identifying this phenological stage is quite complex. Therefore, to avoid confusion and apply GA3 at the most opportune time, it is recommended to do so at the beginning of veraison.
This is based on the understanding that the best time for each phenological stage is the beginning of the next. Thus, the best time for white bud is when the first flowers open, the best time for full bloom is at the beginning of petal fall, and the best time for straw color is at the beginning of veraison.
To recognize the beginning of veraison, in addition to detecting the fruit’s color change from green to yellow, it can be determined when 5-10% of the fruit has begun to turn pink or red. At this point, a large percentage of the fruit on the plant is likely past the color break stage, and the optimal conditions for GA3 application are present.
The exogenous use of GA3 at the beginning of stage 3 of fruit growth is directly related to the final fruit size. However, it should be noted that it is often mistakenly believed that GA3 also increases firmness.
Gibberellic acid, in addition to contributing to cell elongation and fruit size, primarily improves FIRMNESS, since firmness is directly associated with the fruit’s dry matter content. When GA3 is applied, it contributes very little to increasing dry matter, as this develops mainly during the cell division phase, in stage one of fruit growth, perhaps up to 30 days after full bloom.
Now, regarding the concentration of the applications, Avium’s technical team has studied a direct relationship between the concentration of GA3 and fruit size. However, beyond 40 ppm, there is no direct increase in size. Therefore, using more than 40 or 45 ppm generally does not have a greater effect on cell elongation or final size.
The variation in this concentration is subject to several factors, which are explained below:
Orchard load status: General considerations
1. Balanced load status: a medium concentration of GA3 is recommended, i.e., between 20 and 30 ppm.
2. Light load status (below 5,000–6,000 kg/ha): use concentrations around 10 ppm.
3. High load status (over 15,000 kg/ha): use concentrations of approximately 40 ppm.
Cultivars more susceptible to cracking
This aspect is an important factor in deciding on the correct concentration. For cultivars that are more susceptible to cracking, caution should be exercised when using GA3, and the concentration should not exceed 20-25 ppm, as this is a region more prone to cracking. This cracking is generally not caused by rain, but by standing water, dense fog, drizzle, and often condensation in the most humid areas.
Undoubtedly, the benefits of applying this type of phytohormone are numerous, but it should be mentioned that it causes some disruption in fruit color development. This is not a case of “delayed color development,” so in earlier varieties, with the goal of harvesting before November 20-25, excessive use of GA3 is not recommended. In these cases, a concentration of 20-25 ppm is recommended, always assuming it is applied in a single application at the optimal time.
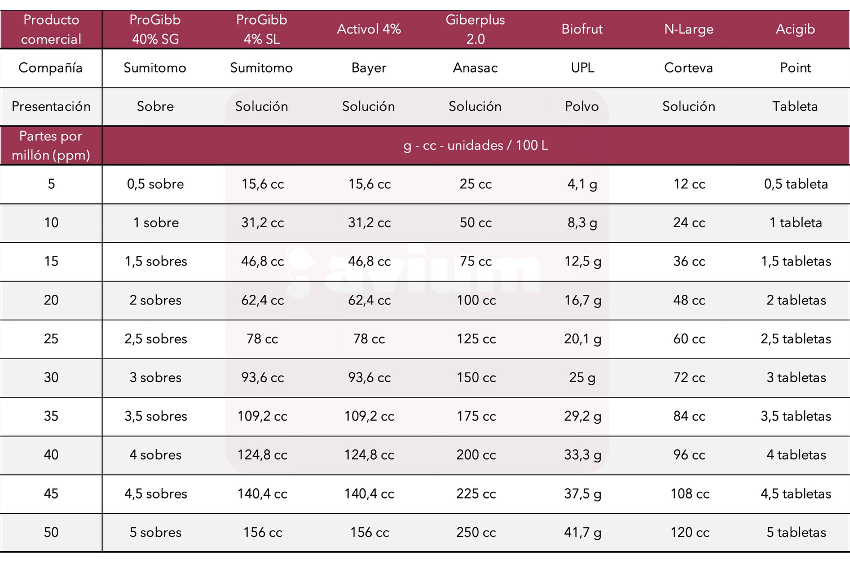
Given the wide variety of GA3 alternatives available in commercial products in Chile, differing in concentration, formulation, and format, Avium has created a table outlining these relationships according to need:
Table 3. Relationships between concentration (ppm) of active ingredient GA3 and concentration (g or cc/100 L) of commercial product available in Chile.
Is GA3 recommended in all cases?
The answer is yes, as this application always has a positive effect. In orchards yielding 2,000 to 3,000 kg per hectare, provided the application method reaches all the fruit (since it works by contact), the minimum concentration to use is 10 ppm.
Furthermore, the exogenous application of phytohormones can be useful in restoring metabolic activities to normal levels. GA3, at a specific concentration, has been shown to be beneficial for the physiology and metabolism of many plants subjected to abiotic stress, as it can regulate the metabolic process based on antioxidant and sugar signaling enzymes.
Regarding the adverse effects of gibberellic acid use, it should be noted that, according to various studies conducted by Avium in recent seasons, when concentrations above 30-35 ppm are used (when justified), pedicel deterioration occurs, causing them to dehydrate more easily.
In these cases, it is recommended to accompany the application of GA3 with some type of plant extract, for example, seaweed extract. This helps to mitigate the potent effect that gibberellic acid has on the elongation of pedicel tissue cells, which are apparently more delicate than those of the fruit.
This prevents these cells from rupturing and subsequently dehydrating. Therefore, GA3 applications exceeding 25-30 ppm should always be accompanied by some type of plant extract.
It is essential to establish the specific concentrations for each situation, as well as to ensure that the application methods are perfect and that it reaches all parts of the plant and 100 percent of the fruit so that it directly relates to the main objective, which is to improve fruit size.
As a practical tip, there is no need to panic when the fruit begins to change color from green to yellow; It will always be better to wait to see the first pink fruits and then apply the best concentration according to the main objective in each case.
References
– Tapia, C., Martínez, E., González, D., Martínez, N., Contreras, M., Morales, M., and Barros, M. 2025. Fruit growth and development analysis in different sweet cherry (Prunus avium L.) cultivars in central Chile. 10th International Cherry Symposium.
– Carrión-Antolí, Á., Serrano, M., Martínez-Romero, D., & Valero, D. (2024). Preharvest gibberellic acid treatments improve firmness and postharvest life of sweet cherries (Prunus avium L.). Scientia Hortícolae, 324, 112678.
– Whiting, M. D., & Lang, G. A. (2021). Use of gibberellic acid (GA₃) to increase fruit size and firmness in sweet cherry (Prunus avium L.). Acta Horticulturee, 1314, 135–142.
– Xu, Y., Zhang, J., & Wang, L. (2025). Effects of gibberellic acid on cracking resistance and fruit physiology in sweet cherry (Prunus avium L.) under different irrigation regimes. Journal of Horticultural Science and Biotechnology, 100(1), 45–56.







